Pharmacodynamics
Pharmacodynamics is the study of the changes and phenomena that a drug produces on the body – its actions/effects, mechanisms of action, receptors, and deleterious effects.
SAQ,Viva
What do you mean by pharmacodynamics?
Pharmacodynamics: (The English word “Dynamic” means one who brings about change, one who makes effect) So, Pharmacodynamics is the study of effects of drug on the body. It includes pharmacological actions/effects of a drug, its mechanism of action, interactions with receptors, adverse effects.
SAQ,Viva
What is the difference between “Pharmacodynamics” and “Pharmacokinetics”?
Dynamics is about the effect of the drug on the body.
Kinetics is about the movement of the drug in the body.
So Pharmacodynamics is the study of effects of the drug on the body systems. This science includes the pharmacological actions of a drug, its mechanism of action, its interaction with receptors, its various effects including the unwanted effects. Whereas, Pharmacokinetics means the absorption, distribution, metabolism, and excretion of the drug. Because in this process, the drug moves in the body, the word “kinetics” (which stands for motion or movement) is used.
Viva
What is the difference between “pharmacological action/effect” or “mechanism of action”?
“Pharmacological actions or effects” – means the changes that the drug produces on various body systems or cells.
“Mechanism of action” means how the drug produces those actions, or what is the way by which the drug produces those actions.
For example: Pharmacological action of salbutamol is to produce bronchodilation, and the mechanism of this action is by stimulating the beta-2 receptors in the bronchial smooth muscles and increasing the cyclic AMP.
Pharmacological actions may be many because they are the effects on different body systems or they are different entities that a drug can do.
For example: Aspirin has many actions like analgesic action (relieving pain), antipyretic action (decreasing body temperature), and antiinflammatory action (decreasing inflammation). But the basic mechanism behind producing all these actions may be just one, and that is – inhibiting prostaglandin synthesis. Thus sometimes actions may be many, but there may be a single mechanism behind these actions.
LAQ
Enumerate the factors modifying drug effect/drug action.
Factors modifying drug action
A. Patient- related factors
- Age
- Body weight/body size
- Sex
- Diet / food
- Species / Race
- Environmental factors/occupation
- Genetic factors
- Presence of disease
- Psychological factors – Placebo
- Psychological factors – Patient compliance
B. Drug- related factors
- Route of administration
- Environment
- Time of administration
- Cumulation
- Tolerance
- Tachyphylaxis
- Drug dependence
- Drug interactions
Viva/SAQ
What are the principles of drug action?
A drug is likely to stimulate or depress a particular system or irritate the cells. A drug may also play a role as a supplement to replace the deficient nutrient in the body. Sometimes a drug may destroy a cell. A drug may work through these five principles.
- Stimulation: Increase in the activity of cell function. For example: If a drug like adrenaline stimulates the activity of cardiac muscle cells, it would lead to stimulation of heart. So adrenaline is called a cardiac stimulant.
- Depression: It is the decrease in the activity of cell function. For example if a drug like diazepam depresses the activity of individual cell function in the Central nervous system (CNS), then the drug is causing CNS depression, and hence called a CNS depressant.
- Irritation: is a noxious effect. Mild irritation may lead stimulation of cell function, but stronger irritation may lead to cell death.
- Replacement: Filling up deficiency of a hormone or an enzyme or a body nutrient e.g. Vitamin B12 to fill up its deficiency, Iron to fill up iron deficiency, thyroid hormones to correct hypothyroidism
- Cytotoxic action: Selective cytotoxicity means cell damaging effect exerted selectively on parasite cells or bacteria or cancer cells to destroy them without affecting the human host cells.
- Inhibition/Blockade of an enzyme/receptor/pump/channel: A drug may act by inhibiting an enzyme/a transporter/an ion channel/a receptor. Sometimes a term blockade/antagonism may be used in place of inhibition, eg. blockade of a receptor/an enzyme/ channel/ pump/ transporter. Such a drug may be called inhibitor or blocker.
- Activation/Stimulation of a receptor/enzyme/pump/channel: A drug may produce activation or stimulation of receptor/ enzyme/ pump/ channel. For a receptor, a drug may then be called as agonist or stimulant.
SAQ/LAQ/Viva
Enumerate with examples various mechanisms of action of drugs
Drugs act by following main mechanisms:
- Physical Mechanism: Mechanism of action of drug is its physical property: Examples
|
Drug |
Physical Mechanism |
Action |
|
Tannic Acid |
Astrigent & adsorbent |
Protects oral ulcer |
|
Mannitol |
Osmotic mechanism |
Diuretic action |
|
Methylcellulose |
Forms bulk |
Laxative |
2. Chemical Mechanism: Drug acts by chemical action: Examples
|
Drug |
Chemical Mechanism |
Action |
|
Aluminium Hydroxide, Magnesium Hydroxide |
Alkali neutralizes the acid |
Antacid action in stomach |
|
Potassium Permanganate |
Oxidizing action |
Kills bacteria by nascent oxygen |
|
Desferrioxamine |
Chelation |
Antidote for iron toxicity |
3. By acting on certain target molecules in body
(a) On Receptors: Examples
|
Drug |
Receptor & Action |
Effect |
|
Propranolol |
Blocks beta receptors |
Decreases heart rate |
|
Atropine |
Blocks muscarinic receptors of acetylchyoline |
Blocks the effects of acetylcholine; thus useful in organophosphorous poisoning |
|
Salbutamol |
Stimulates Beta-2 adrenergic receptor to increase cyclic AMP |
Dilates the bronchial smooth muscle; thus useful in bronchial asthma |
(b) On Enzymes: Examples
|
Drug |
Enzyme & Drug action on Enzyme |
Effect |
|
Pantoprazole |
Inhibits H+K+ATPase Enzyme |
Inhibits gastric acid secretion |
|
Ramipril |
Inhibits Angiotensin Converting Enzyme (ACE) |
Decreases blood pressure |
(c) On Ion Channels: Examples
|
Drug |
Ion channel and action on ion channel |
Effect |
|
Nifedipine |
Blocks L-type calcium channels in blood vessels |
Produces vasodilation and decreases blood pressure |
|
Ethosuximide |
Blocks T-type calcium channels in the CNS |
Useful in petit mal epilepsy (absent seizures) |
(d) On Transporters: Examples
|
Drug |
Transporter and drug action on transporter |
Effect |
|
Fluoxetine (is an SSRI – selective serotonin reuptake inhibitor) |
Inhibits the transporter called SERT (Serotonin Transporter) and inhibits selectively the reuptake of Serotonin |
Concentrates serotonin and helps to relieve depression (acts as antidepressant) |
|
Furosemide (a loop diuretic) |
Inhibits Na+K+2Cl– transporter (sodum potassium 2-chloride transporter-symporter) |
Increases sodium, potassium, chloride excretion in urine, and acts as a diuretic |
SAQ/Viva
Define a receptor. Enumerate/Mention the various types of receptors with examples.
Receptor is a site on the cell membrane or inside the cell (within cytoplasm or nucleus), with which a drug/nutrient specifically combines to initiate/produce specific action from the cell. The receptor is a dynamic entity, and it is a chemical substance of varied nature. It may be a polypeptide (protein), an enzyme or an ion channel or any other form of a chemical.
Types of receptors:
1. G-protein coupled receptors (GPCR): These act through one of the following second messengers which further carry out the function. (Beta receptors, Alpha receptors, Histamine receptor)
- Adenylyl cyclase activation and increase in cyclic AMP
- Phospholipase C: Inositol Phosphate-3,Diacylglycerol (PL_C:IP3-DAG)
- Ca or K channel regulation
2. Intrinsic Ion Channel Receptors: Receptor is associated with an intrinsic ion channel.
- Nicotinic Cholinergic receptor (Na+channel)(Skeletal neuromuscular junction)
- GABA-A receptor: Chloride channel
3. Enzyme-linked Receptor: Receptor triggers an enzyme.
- Intrinsic enzyme: Insulin receptor: Tyrosine kinase
- JAK-STAT Kinase: Cytokines/Interferrons
4. Intracellular Receptor: Receptor is not on the cell membrane but is inside the cell.
- Cytoplasmic receptor: Steroid receptor, Vitamin A receptor
- Nuclear receptor: Androgen, Estrogen, Thyroxine, Vitamin A receptor
|
Receptor |
Examples |
|
1. G protein coupled receptor (GPCR)
|
Beta-1, Alpha-2, H-2 receptors |
|
a. Adenylyl cycloase (cAMP) |
|
|
b. PL-C:IP3-DAG |
Alpha-1, H1 receptors |
|
c. Channel regulation |
Ca, K Channels (Heart) |
|
2. Intrinsic Ion Channel |
Nicotinic receptor: (Na+ Channel) GABA-A receptor: (Chloride Channel) |
|
3. Enzyme-linked receptor |
|
|
a. Intrinsic Enzyme |
Insulin receptor (Tyrosine kinase) |
|
b. JAK-STAT Kinase |
Cytokines, interferrons receptor |
|
4. Intracellular receptors |
|
|
a. Cytoplasmic receptor |
Steroid receptor, Vitamin D receptor |
|
b. Nuclear receptor |
Thyroxine, Estrogen, Androgen receptor, Vitamin A receptor |
SAQ/Viva
Explain the terms – Affinity, Intrinsic Activity, Agonist, Antagonist, Partial Agonist, Inverse Agonist
Affinity: Capacity of a drug to combine with a receptor
Intrinsic Activity (Efficacy): Capacity of a drug to initiate action from the receptor (after combining)
Agonist: is the one who has affinity as well as intrinsic activity
Antagonist: is the one who has affinity, but has poor / no intrinsic activity.
Partial Agonist: is the one who has equal affinity as that of the agonist, but intrinsic activity is less than that of the agonist.
Inverse Agonist: has equal affinity with that of the agonist, but has negative intrinsic activity.
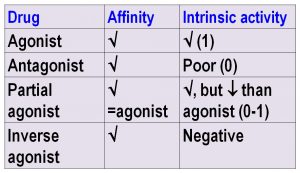
Agonist is many times called a stimulant. For example, Salbutamol is a selective Beta-2 receptor stimulant or a Beta-2 agonist.
Antagonist is many times called a blocker. For example, Tamsulosin is an alpha receptor antagonist or an alpha blocker.
SAQ/Viva
What is” Down-regulation” of receptors? Write its implications.
–Downregulation of receptor is gradual decrease in receptor number, caused by a constant , long-term use of an agonist.
-Agonist USES the receptor –> DECREASE in receptor number –> Gradual DECREASE in response
- Destruction of receptor or DECREASED synthesis of receptor causes slow decrease in response.
Therefore long-term use of AGONISTS often can cause slow decrease in response over the long period of time.
Clonidine is a central alpha-2 agonist, and when it combines with this receptor, it decreases blood pressure because the receptor is inhibitory. But chronic use of clonidine leads to downregulation of receptors. So if clonidine is abruptly stopped, because the receptor number is less, there is an opposite effect, and the blood pressure suddenly rises.
SAQ/Viva
What is” Up-regulation” of receptors? Write its implications.
When an antagonist or a receptor blocker is used over long period of time, thought the drug combines with the receptor, it does not use the receptor. So there is a gradual increase in receptor number or increase in sensitivity of the receptor.
When such drug is abruptly stopped, it can lead to a sudden, high-grade response.
Long term use of an antagonist / blocker –> Receptor NOT USED –> INCREASE in receptor number / receptor sensitivity.
So Sudden stoppage of antagonist –> Supersensitivity and sudden response
Example: ß blockers à Long term use and sudden stoppage leads to Angina / Increased blood pressure.
SAQ/Viva
Explain the term – ADDITIVE EFFECT with appropriate examples.
The term ADDITIVE EFFECT is used when two drugs with similar actions are used together, and they produce addition of individual effects, leading to a higher effect.
Additive Effect: Arithmetic addition of effects of two drugs
(AB)= (A) + (B)
Effect of combination AB = (Effect of A) + (Effect of B)
Examples:
1. Aspirin + Paracetamol = Addition of analgesic effects
Aspirin and paracetamol both relieve pain (analgesic action). So if they are used together, they produce additive analgesic effect
2. Diazepam + Diphenhydramine = Addition of CNS depressant effects
Diazepam is a sedative hypnotic, which depresses the central nervous system. Diphenhydramine is a sedative antihistamine drug, which also produces CNS depression. So using these two drugs together produces addition of individual effects, and may produce severe CNS depression.
Similarly Alcohol + diazepam if used together will produce additive CNS depression, because both the drugs are CNS depressants.
SAQ/Viva
Explain the term – SYNERGISM / SUPRA-ADDITIVE EFFECT with appropriate examples.
The term SYNERGISM or DRUG SYNERGISM or SUPRA-ADDITIVE EFFECT is used when two or more drugs mostly with similar actions when used together, they produce beneficial effect which is even more than just the arithmetic addition of individual effects of the drugs.
Synergism: Effect of the combination is more than just the Arithmetic addition of effects of two drugs
(AB) > (A) + (B)
Effect of combination AB > (Effect of A) + (Effect of B)
Examples:
1. A combination – Cotrimoxazole, which is – Sulfamethoxazole + Trimethoprim.
Sulfamethoxazole and Trimethoprim – individually are bacteriostatic, but the combination is bactericidal. Sulfamethoxazole and trimethoprim produce sequential blockade in the bacteria by inhibiting PABA reductase and Folate reductase respectively. Also, in the combination, the doses of individual drugs needed are less, and the duration of action of the combination is better than that of the individual drugs.
2. Estrogen-progestin oral contraceptive combination.
3. Nitrate-Propranolol combination used for prophylaxis of angina.
4. Hydrochlorothiazide plus Enalapril combination for hypertension.
Synergism may also exist in the form of combinations of two drugs, wherein both drugs need not have similar effects, but one of the drugs may facilitate or help or potentiate the effect of the other drug.
Examples:
- Levodopa-carbidopa combination: Carbidopa – a dopa decarboxylase inhibitor, inhibits peripheral breakdown of levodopa and facilitates its entry into brain, to increase the availability of dopamine, which is useful in treatment of Parkinson disease.
- Lidocaine – Epinephrine combination: Epinephrine constricts the blood vessels and decreases the rate of entry of lidocaine, and thereby prolongs the duration of local anesthesia.
- Amoxicillin – Clavulanic Acid combination: Clavulanic acid – A beta lactamase inhibitor, prevents destruction of amoxicillin by beta lactamase producing bacteria, and thus retain the action of amoxicillin.
SAQ/Viva
Explain the term – DRUG ANTAGONISM. Mention the various types of antagonism with appropriate examples.
DRUG ANTAGONISM: is a phenomenon in which two drugs produce opposite effects to each other. The opposition of effects may happen by any mechanisms.
Based on the mechanism involved, the types or classification of drug antagonism can be viewed as follows:
A. RECEPTOR ANTAGONISM (PHARMACOLOGIC ANTAGONISM)
Two drugs act on the same receptor. This is further divided into:
1. Competitive Antagonism: Two drugs art on the same receptor, and the same site on this receptor. The antagonist blocks the receptor. To reverse this effect, increasing doses of agonist are needed. Thus the original dose-response curve of the agonist gets shifted to the right. The blockade by antagonist can be reversed by gradual increase in the dose of the agonist; thus the agonist can gradually and finally displace the antagonist from the receptor site. to obtain its maximal effect. Thus, because this antagonism can be reversed, it is also called “Reversible or Surmountable Antagonism.”
Examples:
- Diazepam– Flumazenil
- Morphine- Naloxone

2. Non-competitive Antagonism: Two drugs act on the same receptor, but on a different site on the receptor. (an allosteric site). The antagonist combines with the allosteric site on the same receptor, and produces a change in the site where the agonist is supposed to combine. So the agonist cannot easily combine with the receptor site. This antagonism is difficult to reverse, and hence called “Irriversible or Non-Surmountable Antagonism”.
Examples:
- Nor-epinephrine-Phenoxybenzamine
- Diazepam- Bicuculline
- Glutamate – Ketamine
B. NON-RECEPTOR ANTAGONISM (NON-PHARMACOLOGIC ANTAGONISM) (FUNCTIONAL ANTAGONISM)
Here the two drugs have opposite effects, but the mechanism is not related to the same receptor. The two drugs need not combine with the same receptor to produce the opposite effects. They may act by different mechanisms. Hence called “Non-receptor antagonism” or “Non-Pharmacologic antagonism”. Based on the 3 different mechanisms, non-receptor antagonism is further divided into:
1. Physiological Antagonism: The two drugs act on the same system / tissues) , and produce opposite actions.
Examples:
- Epinephrine- Histamine: Epinephrine increases blood pressure, while histamine decreases blood pressure. Epinephrine dilates the bronchial smooth muscle, while histamine constricts the bronchial smooth muscle to produce bronchospasm. Epinephrine acts on its own adrenergic receptors, and histamine acts on its own histaminergic receptors. So they don’t act on the same receptor, but produce opposite effeccts on the same tissue/system. Hence it is physiological antagonism.
- Glucagon – Insulin: Both act on different receptors but glucagon increases the blood glucose, while insulin decreases it.
- Hydrochlorothiazide (a thiazide diuretic)-Triamterene (potassium sparing diuretic): Hydrochlorothiazide decreases potassium levels and triamterene increases the potassium levels; both acting by their own mechanism.
- Aluminium Hydroxide-Magnesium Hydroxide: Aluminium hydroxide decreases intestinal motility, while magnesium hydroxide increases the intestinal motility. Both produce physiological antagonism which is beneficial because they nullify the effects of each other, when they are combined as antacids.
2. Chemical Antagonism: a chemical reaction between two drugs to produce opposite effect.
Examples:
- Gastric Hydrochloric acid – Magnesium hydroxide: Magnesium hydroxide is an alkali, which neutralizes the gastric HCl, and thus produces antacid effect.
- Barbiturate – Sodium Bicarbonate: Sodium bicarbonate (an alkali) given intravenously increases the ionization of the barbiturates (acidic in nature), and thus facilitates its elimination. Hence sodium bicarbonate is useful in barbiturate poisoning.
- Iron-Desferrioxamine (Chelation): Desferrioxamine (Desferoxamine) combines with iron to form an inactive complex (chelate complex), and thus nullifies its effect, and facilitates its elimination, hence useful in iron toxicity.
3. Physical Antagonism: a drug nullifies the effect of another drug by a physical phenomenon/reaction.
Example:
- Activated charcoal – Alkaloidal poisoning: Activated charcoal can “adsorb” the remaining alkaloid in the stomach, when used for gastric lavage in alkaloidal poisoning, And thus helps to remove the unabsorbed poison from the stomach. The physical phenomenon involved is called “Adsorption”.
SAQ/Viva
Define “cumulation” with examples.
Cumulation is the process of deposition of a drug in particular tissues. Drugs that have high volume of distribution (non-homogenous distribution) have high affinity for certain tissues and a tendency to get deposited into those tissues. Such drugs may produce beneficial effects on those tissues. So also, there is a risk of toxicity to these tissues due to drug cumulation. Increased frequency of administration, administration in higher dosages, and prolonged administration shall obviously increase the susceptibility for adverse effects of such drugs.
Examples
- Tetracyclines – Teeth, Bones
- Chloroquine – Retina, liver
- Streptomycin – Vestibular apparatus
- Emetine, digoxin – Heart
- Iodine, Amiodarone – Thyroid, lungs, skin, liver
- Dapsone, Clofazimine – Skin, Reticulo Endothelial System [RES]
SAQ/Viva
Define Adverse Drug Reactions/Adverse Effects of Drugs and enumerate the main types.
Adverse effects of drugs / Adverse Drug Reactions (ADRs) are the unwanted effects of drugs usually produced in therapeutic doses. They are the noxious harmful (noxious), unintended effects of a drug when a drug is used in usual doses for its various indications.
They are classified in to 5 types:
Type A, B, C, D, E
SAQ/LAQ/Viva
Classify adverse drug reactions with examples
|
Type – Name |
Description / Examples |
|
A – Augmented (Dose-related) |
Extension of pharmacological action B blockers – Bradycardia Atropine – Dry mouth Glipizide – Hypoglycemia Warfarin – Bleeding |
|
B – Bizarre (Qualitative) (Non-dose related) |
-Hypersensitivity/allergy: Immunological reaction -Idiosyncrasy: Non-immunologic, other mechanisms, may have genetic basis |
|
C – Continuous reactions |
-Due to long term use -Steroids – Cushing syndrome -Analgesics – Nephropathy |
|
D – Delayed reactions |
Carcinogenicity, Mutagenicity, Teratogenicity |
|
E – End of use reactions |
WITHDRAWAL / DISCONTINUATION of a drug after long term use – Steroids – Acute adrenal insufficiency Beta blockers – Sudden hypertension or precipitation of angina Barbiturates – Rebound REM Benzodiazepines – Insomnia, anxiety, seizures |
SAQ/Viva
Define “Teratogenicity” with examples.
Teratogenicity is the capacity of a drug to produce structural defects or deformities in the fetus, when a drug is administered to a female during pregnancy.
Terat means a monster or a structurally strange object, hence the word “teratogenicity” should be used specifically to describe the structural defects or deformities or abnormalities, and not in general to every abnormality produced in a fetus.
In general, the process of organogenesis takes place during the early pregnancy (upto first 8 weeks or 55 days). Hence the term “teratogenicity” applies to the drug administration during this period.
Naturally, teratogenicity is the manifestation of the drug administration during the first trimester of pregnancy.
Teratogenic effects: Examples
- Thalidomide – Phocomelia – seal limbs – short limbs
- Phenytoin – Neural tube defects including spina bifida, anencephaly
- Warfarin – Depressed nose; Eye, hand defects
- Tetracyclines – Teeth abnormalities including discolored, weak, fragile, and ill-developed or carious teeth.
SAQ/Viva
Enumerate some harmful effects of drugs on fetus, when the drugs are administered during 2nd or 3rd trimester of pregnancy. /Enumerate some non-teratogenic effects of drugs.
When the drugs are administered during 2nd or 3rd trimester of pregnancy, some drugs may lead to certain complications. These are not structural defects, hence they are not called “teratogenic” effects.
Such non-teratogenic effects include other complications.
Examples:
- Warfarin – fetal hemorrhage
- Morphine – respiratory depression
- Oral antidiabetics – hypoglycemia
- Lithium – fetal goiter
- ACE inhibitors – hypoplasia of organs
SAQ/Viva
Compare and contrast: Type A and Type B Adverse Drug Reactions / Write differences between Type A and Type B Adverse Drug Reactions.
A stands for “Augmented”. “Augment” means to increase in number/amount. So Type A adverse drug reactions are those which happen due to administration of more quantity/dosage of drug, and they are usually extension of the pharmacological actions; naturally they are predictable. So they are mostly quantitative or dose-related.
B stands for “Bizarre”. “Bizarre” means very strange or unusual. So, Type B adverse drug reactions are unexpected responses even to smaller doses of drugs. They don’t happen commonly or in most of the individuals. They may not be related to the dose. They are mostly qualitative.
|
Type A ADRs: (Augmented) |
Type B ADRs: (Bizarre) |
|
Due to increased doses (Dose related) |
Can happen with a single dose (Non-dose related) |
|
Quantitative |
Qualitative |
|
Extension of known pharmacological action of a drug |
Not based on action of a drug; may be related to the peculiar conditions present in a patient |
|
Based on the mechanism of action of that drug |
Mechanism may not be known |
|
Examples: Hypotension due to calcium channel blockers Bradycardia due to beta blockers |
Examples: Idiosyncrasy to chloramphenicol, Hypersensitivity to penicillins |
|
More common |
Less common |
|
May be less serious, (of course depending upon the dose ingested) |
Generally more serious |
|
May be treatable / reversible / may be controlled by decreasing the drug dosage or stopping the drug |
May be difficult to treat / difficult to reverse / of course shall need stoppage of the drug, but if it is acute, the manifestation already has been much serious |
|
Mostly predictable |
May not be predictable (May be predicted if the genetic basis in a particular individual is known) |
|
May be preventable |
May not be easily preventable, unless there is prior knowledge of the phenotype or genetic basis |
SAQ/Viva
What is Iatrogenic disease? Give examples
Iatrogenic disease is the disease condition produced due to administration of drug treatment. The original Greek word “iatro” means healer or physician or medicine. So, “Iatrogenic disease” is sometimes popularly called as “Drug-induced disease” or “Physician-induced disease”
Examples
|
Iatrogenic Disease |
The drug which is responsible |
|
Cushing syndrome, Diabetes mellitus |
Steroids |
|
Peptic ulcer |
NSAIDs, steroids |
|
Parkinson-like syndrome |
Antipsychotics |
|
Hepatitis |
Isoniazid |
|
Renal failure |
Aminoglycosides, Amphotericin B |
|
Alopecia |
Anticancer drugs |
Although called physician induced, but really speaking, they are drug induced. Because many times, the patients continue the drugs on their own for longer duration, and it leads to a disease. The best example is steroids, which are inadvertently taken.
In other cases, a drug needs to be given to the patient for a particular indication, and then it leads to the disease or harmful effect. Anticancer drug is given with an intention to help in cancer treatment, but it may inevitably produce alopecia as its unwanted effect.
Drugs administered for long duration may have been prescribed intentionally, because they are needed to be given (fully knowing that some harmful effects may happen), but the benefit is going to be more than the harm.
In some other cases, the drug administration for longer period may have been overlooked by the physician due to various reasons, and then the iatrogenic disease is evident.
SAQ/Viva
What is Therapeutic Index? Explain its implications. Enumerate drugs with low therapeutic index and high therapeutic index.
Therapeutic Index (T.I.) is the Index derived by dividing the median lethal dose (LD50) by medial effective dose (ED50).
T. I. = LD50 / ED50
During discovery of a new drug, animal experiments are conducted in which the drug toxicity is studied. During this study, the average lethal dose in animals is calculated. (The dose required to kill more than 50{2610912583dabb07030bf2cdad608afafcc4beb0908ae67f305141b9a5c99c63} of the animal population). This is called LD50. Before this, the average effective dose is calculated. (The dose required to produce expected therapeutic effect in more than 50{2610912583dabb07030bf2cdad608afafcc4beb0908ae67f305141b9a5c99c63} of animal population). This is called ED50.
So, the ratio of lethal dose and effective dose is therapeutic index. So it is the calculation of – “Lethal dose is how many times the effective dose”.
More the therapeutic index, safer will be the drug. Therapeutic index is the index or measure of safety of a drug.
Because more therapeutic index indicates that there is wide difference between the therapeutic dose and killing dose.
The therapeutic index has to be obviously more than 1, because the lethal dose has to be more than the effective dose.
Calculation:
|
Drug A has an ED50 of 5 and an LD50 of 60. Drug B has an ED50 of 10 and an LD50 of 30. Which drug is better and Why? |
|
|
For Drug A T.I.= LD50 / ED50 = 60 / 5 = 12 |
For Drug B T.I.= LD50 / ED50 = 30 / 10 = 3 |
|
Therapeutic Index of Drug A is 12. Therapeutic Index of Drug B is 3. Hence Drug A is better. This is so because there is a wider difference between LD50 and ED50. So Drug A is safer than Drug B. For Drug A, there is a wide margin of safety between which there is a scope of increasing the drug doses. |
|
More the difference between effective dose and lethal dose, more will be the value of the therapeutic index. This means there is wide margin of safety in which one can go on increasing the dose of this drug. So more therapeutic index means more safety.
For practical purposes, therapeutic index is now many times taken as the ratio between the toxic dose and effective dose and then written as –
T. I. = TD50 / ED50
Here, the toxic dose means the minimal dose that starts producing toxic effects. Thus starting from the minimal effective dose the drug dosage can be adjusted until the minimal toxic dose at which the drug produces adverse effects. This is the real therapeutic window (margin) of safety. Wider the therapeutic margin, safer is the drug. Thus the difference between these two doses provides a range between which the drug dosages can be altered or adjusted.
|
Drugs with fairly good or better T.I. |
Drugs with low T. I. (Less safe drugs) |
|
Penicillins, Cephalosporins, macrolide antibiotics -Aspirin, Ibuprofen -Propranolol, Telmisartan -Remifentanyl |
Morphine, Barbiturates, Cocaine, Ethanol, Carbamazepine, Lithium, Phenytoin, Fosphenytoin Digoxin, Digitoxin, Procainamide Tetracyclines, Chloramphenicol, Aminoglycosides (Gentamicin, Streptomycin), Clindamycin, vancomycin, Amphotericin B, Anticancer drugs Tacrolimus, Sirolimus Warfarin, Theophylline |
SAQ/Viva
Enumerate the Pregnancy risk categories with examples, and write their interpretation.
Pregnancy risk categories describe the severity of risk of the drugs to the fetus, if the drug is administered during pregnancy.
The categories are based on the evidence from observations and experiences in pasat from clinical set-up and from the observations in the experimental animals.
From A to X, the risk goes on increasing. Drugs in category A are supposed to be the most safe, and as you go further down until the category X, the risk is highest.
For example – Penicillins and other beta lactam antibiotics are comparatively safer whereas tetracyclines, chloramphenicol, and most of the aminoglycosides and antiepileptics are toxic to the fetus.
| Risk Category | Examples |
| A | Injection MgSO4, thyroxine |
| B | Beta lactams (Penicillin V, amoxicillin, cefaclor), clindamycin, erythromycin, azithromycin, metronidazole, sulfa drugs, paracetamol, lignocaine |
| C | Chloramphenicol, fluoroquinolones, clarithromycin, trimethoprim, vancomycin, gentamicin, cotrimoxazole, morphine, codeine, atropine, steroids, thiopentone, bupivacaine, adrenaline, gabapentin |
| D | Tetracyclines, all aminoglycosides (except gentamicin), aspirin, phenytoin, carbamazepine, valproate, lorazepam |
| X | Estrogens, isotretionin,ergometrine |